Research in the Webb lab is highly diverse, with projects that often sit at the interface of chemistry and molecular biology. Students in my lab will have the opportunity to learn a variety of experimental techniques, while working in a collaborative and fun environment. This allows for every chance of success, while learning fundamental skills that are important for any future career path. The three main research areas in the Webb group are listed below.
1) Ruthenium Complexes for Alzheimer's Therapy
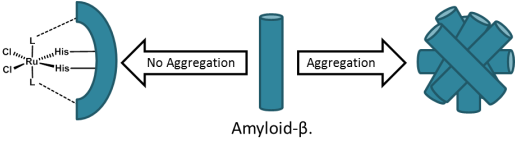 Inhibition of amyloid-beta aggregation by Ru(III) complexes.
Inhibition of amyloid-beta aggregation by Ru(III) complexes.
Protein aggregates are one of the hallmarks of neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease. For Alzheimer’s, Amyloid-β (Aβ) plaques are dense deposits of proteins and cellular matter which accumulate around nerve cells have been observed to have an affinity for metal ions such as copper, iron, and zinc. One promising research area is the inhibition of such protein aggregates in solution, whereby complexes bind to Aβ monomers, and prevent subsequent aggregation. Previous research has focused on using platinum-based complexes where the metal center coordinates to histidine residues within the Aβ peptide, while aromatic ligands on the metal promote non-coordinate, hydrophobic, interactions with the peptide. Recently, Ru(III) anticancer complexes and their derivatives have been re-evaluated for their ability to prevent Aβ aggregation. Such compounds maintain their original octahedral geometry around the ruthenium metal center, and resemble the “Keppler-type” family of Ru(III) anticancer complexes. Interestingly, the complexes which displayed low anticancer activity were found to have a significant effect on reducing aggregation. Similar to their platinum predecessors, the complexes were observed to bind both directly to the peptide, coordinating to histidine residues, and indirectly, through hydrophobic interactions. With this in mind, we are proposing to synthesize complexes which should have an affinity for hydrophobic protein interactions, while also having exchangeable ligands to facilitate protein coordination.
2) Antibacterial Ruthenium Complexes
Metal-based complexes represent an auspicious avenue for antibacterial development, as the coordination environment around the metal center offers stereochemical variability not available with conventional organic complexes. Furthermore, although DNA has been a central target for such complexes given their common positive charge and resultant electrostatic interactions with DNA, alternative targets are also common with proteins and cell membranes have also been the primary target for metallotherapeutics. Ruthenium-based therapeutics have seen significant success as anticancer agents; however, their exploration as antibacterial candidates has not seen the same interest. Therapeutic ruthenium(III) complexes typically have an octahedral geometry with coordinated heterocyclic ligands or a dimethyl sulfoxide (DMSO) in the axial positions, while an equatorial plane of chlorides completes the coordination sphere. These complexes can be further divided into two distinct families, based on the nature of the coordinating axial ligands, and lead compounds that have undergone clinical evaluation. For therapeutic Ru(II) complexes, the ligands around the metal center are commonly arranged in a piano-stool configuration. Here, one face of the complex is coordinated to an arene, while the other face has either a mono- or bidentate ligand with the remaining coordination sites occupied by chloride ligands. Again, there are two distinct families of compounds for Ru(II) complexes, depending on the nature of the coordinating ligand. Building upon the anticancer success of ruthenium therapeutics, in the Webb research group we will look to expand the therapeutic utility of ruthenium complexes as antibacterial agents where derivatives of the previously successful anticancer agents will be prepared and evaluated.
3) Iron and Parkinson's Disease
Parkinson’s disease is characterized by the presence of protein aggregates called Lewy bodies. These plaques are primarily composed of oligomers of the protein α-synuclein (αS), which is a small protein of 140 amino acids that is natively unfolded. The folding of this protein has been found to be accelerated in the presence of metal ions, while elevated amounts of iron have been found within the brains of individuals diagnosed with Parkinson's disease. Furthermore, relatively high levels of iron have been observed within Lewy bodies, which leads to the generation of reaction oxygen species (ROS) is observed, which attributes to the toxicological effects of the Lewy bodies. Our research focuses on preparing new iron chelators as a potential therapeutic strategy, where a hallmark of the disease is targeted directly.
